Dr Minh Alexander retired consultant psychiatrist 17 April 2023
A key matter which triggered BBC Newsnight reportage of the governance failings at University Hospitals Birmingham NHS Foundation Trust was the scandalous whistleblowing case of Tristan Reuser, ophthalmic surgeon.
Mr Reuser was seriously mistreated and unfairly dismissed.
One of the key issues was that he was unfairly suspended based on an unfounded allegation that the ET determined that Clive Ryder Deputy Medical Director and David Rosser Medical Director, later Chief Executive, knew or ought to have known was false.
A related, central issue was that the trust failed to show Tristan Reuser correspondence from an NHS agency called the National Clinical Assessment Service, NCAS (now called Practitioners Performance Advice, PPA), which has a statutory role in advising employers on the suspension of and disciplinary action against doctors.
The withheld correspondence would have revealed that his suspension was triggered on a false premise.
NHS employers had to consult NCAS as part of triggering the euphemistically named “Maintaining High Professional Standards in the Modern NHS” (MHPS) disciplinary and capability procedure.
The MHPS rules of procedures require NHS employers to show referred doctors all correspondence relating to their case.
UHB not only failed to show Tristan Reuser the NCAS correspondence that he was entitled to see, but when Tristan Reuser asked for his personal data, UHB failed to disclose the NCAS correspondence.
The resistance to share the NCAS correspondence persisted into Employment Tribunal proceedings when UHB failed to disclose material documents even in the legal disclosure process, incurring the judge’s displeasure and a £20,000 costs award against the trust.
The effect of UHB’s secrecy was that misleading information supplied to NCAS by Clive Ryder UHB deputy medical director, to justify Tristan Reuser’s suspension, was hidden.
How could this have happened in a process that is supposedly safeguarded?
An MHPS “Designated Board Member” was allocated to Tristan Reuser’s case: Jon Glasby, non executive director, who was appointed as the UHB Independent Senior Director under Jacqui Smith’s tenure.
The Designated Board Member’s role in MHPS is to ensure fair play and that the accused doctor is not “incarcerated” by default through an excessively drawn out process.
“The Chairman of the Board must designate a non-executive member “the designated member” to oversee the case and ensure that momentum is maintained.”
“Role of designated Board member
13. Representations may be made to the designated Board member in regard to exclusion, or investigation of a case if these are not provided for by the NHS body’s grievance procedures. The designated Board member must also ensure, among other matters, that time frames for investigation or exclusion are consistent with the principles of Article 6 of the European Convention on Human Rights (which, broadly speaking, sets out the framework of the rights to a fair trial).”
During his phase one investigation at UHB, Mike Bewick was told that the UHB consultant body had raised concerns with the UHB board about cronyism at all levels of medical management.
He was also told of staff perceptions of arbitrary discipline at the trust.
As a former Deputy Medical Director himself, he would surely understand the requirements of MHPS and the significance of any breaches of procedure.
In his report, Bewick stated that he considered that the findings of the Employment Tribunal in Reuser v UHB and the GMC’s warning to David Rosser called into question David Rosser’s fitness for a senior leadership role:
“31. In our opinion, statements made by the GMC and the Employment Tribunal were extremely serious and brought into question DR’s suitability for senior leadership roles.”
It follows that Rosser’s critical actions as a medical director, in suspending and disciplining doctors, should also be reviewed.
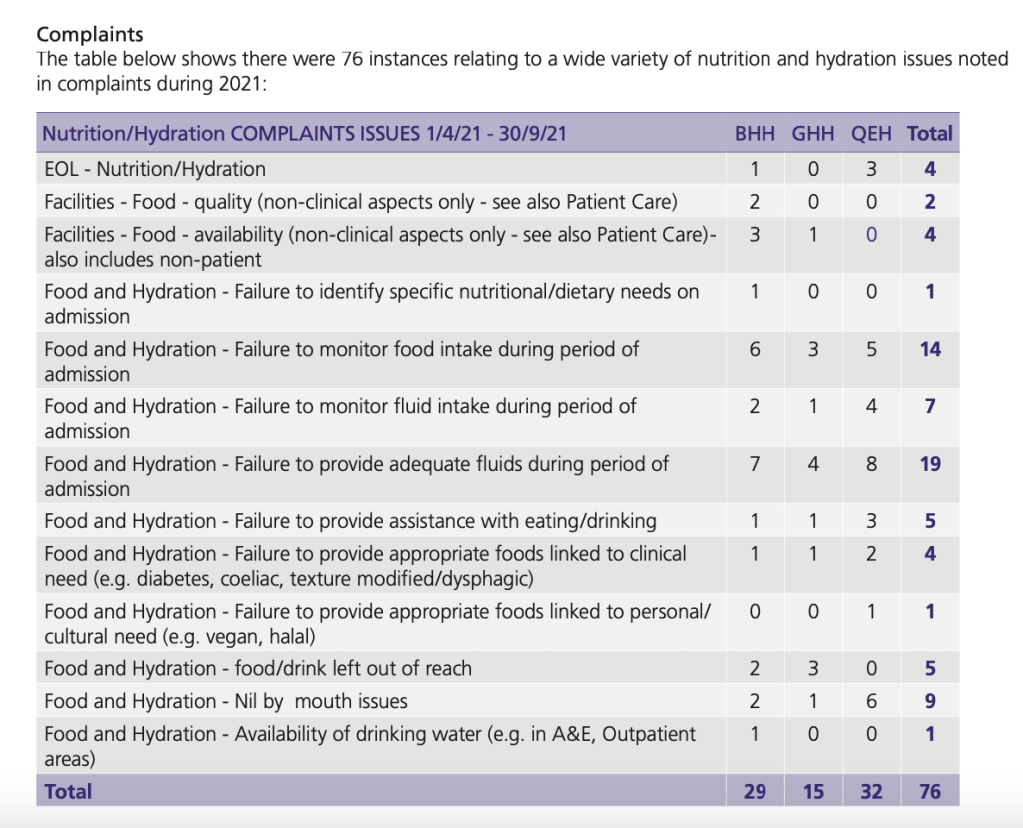
As part of Bewick’s investigation, could he not have called up data on UHB’s use of MHPS and adherence to the rules of procedure?
There was anecdote in Bewick’s report about this, but no hard data, making it inconclusive:

Dragging out the review process may benefit the UHB gravy train but not staff and patients.
In the meantime, I have asked the interim UHB Chair to consider some basic, urgent checks.
I have also suggested that when MHPS is applied at UHB, doctors should be clearly informed of their right to access case correspondence.
BY EMAIL
Yve Buckland
Interim Chair
University Hospitals Birmingham NHS Foundation Trust
17 April 2023
Dear Yve,
Failures of MHPS process
I do not know how familiar you are with the NHS conduct/capability process for disciplining doctors, Maintaining High Professional Standards in the Modern NHS (MHPS), but it has rules of procedure with supposed safeguards.
These failed entirely in the case of Tristan Reuser, UHB whistleblower.
One of the MHPS rules is that doctors who are subject to these procedures must be allowed to see all correspondence about their case, to fully enable them to reply to all allegations.
UHB not only failed to ensure this access to case correspondence, but persistently withheld crucial correspondence from Mr Reuser. This withheld correspondence showed that Clive Ryder Deputy Medical Director gave misleading information to an NHS body the National Clinical Assessment Service, as part of the procedure of triggering suspension under MHPS rules. This was later criticised by the Employment Tribunal as “beyond inaccurate”.
Was it really the case that the then deputy medical, the medical director, the director of human resources and the MHPS designated board member who should have ensured fair play (Jon Glasby), all experienced directors, were not aware that Mr Reuser should have been given sight of his MHPS case correspondence?
My concern now is how many other UHB doctors have been disadvantaged in this way?
How many staff are currently being disadvantaged in this way?
I have looked at the UHB MHPS policy.
This document lists the many alleged failings that may land a doctor in hot water. But it does not say much about the organisation’s responsibilities to treat an accused doctor fairly.
Importantly, I cannot see that the policy says anything about an accused doctor’s rights to see all case correspondence.
The UHB policy cross references the national MHPS framework but gives no link.
It would empower staff if UHB actively gives access to the detailed rules of procedure. MHPS is arguably a contractual matter, and a matter of rights.
The national framework is a lengthy document and the information about the right to see case correspondence is brief and easily missed.
Page 11 of the national MHPS framework:
“3. The practitioner concerned must be informed in writing by the case manager, as soon as it has been decided, that an investigation is to be undertaken, the name of the case investigator and made aware of the specific allegations or concerns that have been raised. The practitioner must be given the opportunity to see any correspondence relating to the case together with a list of the people that the case investigator will interview. The practitioner must also be afforded the opportunity to put their view of events to the case investigator and given the opportunity to be accompanied.”
It would be helpful if the local UHB policy spells out clearly the right of access to case correspondence.
Does UHB need to rapidly audit adherence to basic MHPS standards, based on audit standards co-produced with staffside, to ensure that no further serious harm to individuals is currently unfolding, and that any other serious harm that has taken place besides Reuser v UHB is corrected?
With best wishes,
Minh
Dr Minh Alexander
Cc Chaand Nagpaul External reference group
RELATED ITEMS
Waste Industry: The NHS disciplinary process & Dr John Bestley
Bewick, the ICB, misinformation by UHB about GMC referrals and a late correction
Mr Tristan Reuser’s whistleblowing case: Scandalous employer and regulatory behaviour on FPPR
NHSE, ICB and UHB’s three-ring circus and Rosser’s digital assignment
Can Healthwatch Birmingham please let us see its complaints & concerns data on UHB?
Bewick’s silence on patient complaints at University Hospitals Birmingham NHS Foundation Trust